The BioProtect Balloon Spacer Procedure
Workflow-efficient, image-verified placement.
The BioProtect placement procedure fits into existing radiation treatment preparation workflows. The procedure is performed immediately after placement of the fiducial markers using the transperineal technique,1 and BioProtect’s visibility on TRUS, MRI, and CT supports verification of consistent placement.2
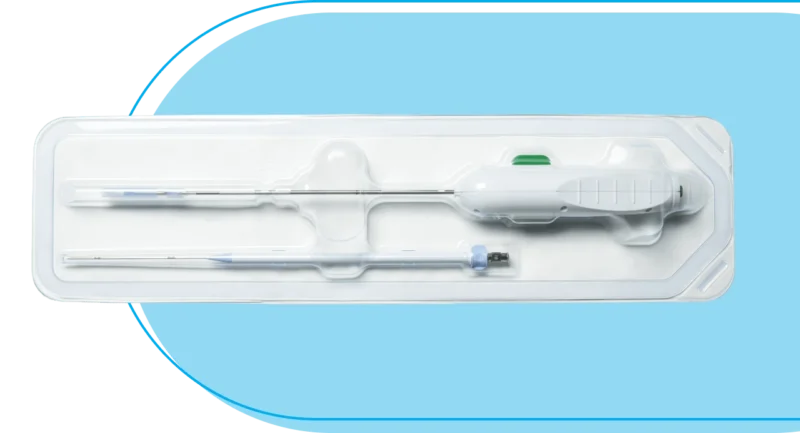
Placement Procedure
BioProtect placement is performed and verified in six steps.
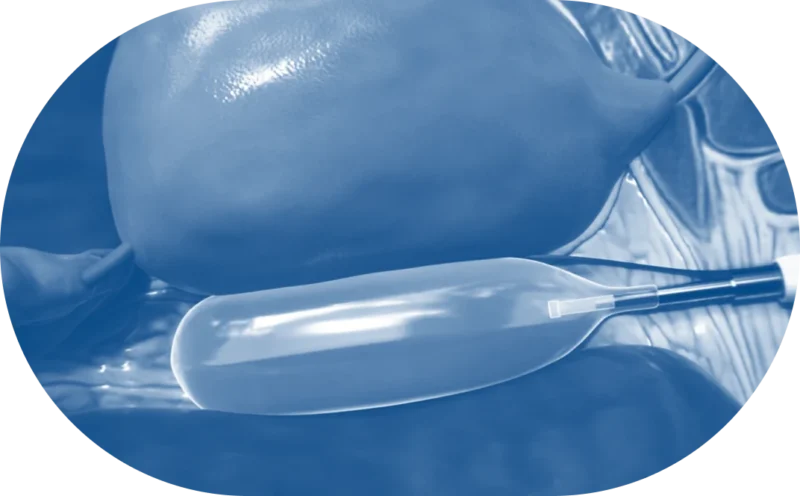
Predictable Workflow, Familiar Equipment
Required equipment to perform the BioProtect procedure includes:
- BioProtect Balloon Spacer kit.2
- Saline solution (0.9%).2
- 20 ml sterile, disposable luer-lock syringe.2
- Scalpel.2
- 10 ml tapered/slip tip syringe.2
- Suturing kit.2
- Side-fire trans-rectal ultrasound (TRUS).
- Stepper/stabilizer.
Learn More About the
BioProtect Balloon Spacer
Get in touch with a BioProtect representative to explore additional information about the BioProtect procedure.
